|
6/21/2023 0 Comments Hydrogen bonds In several cases intermolecular H⋯F bridges dominate, and play a significant role in packing motifs, even when the underlying molecules tend to form only intramolecular bridges in solution. Some database evaluations, however, have shown enough cases for interactions between fluorine and X–H donors, mostly with H⋯F distances around 2.5 Å and X–H⋯F angles around 130°, which makes it difficult to distinguish hydrogen bonds from dispersive interactions. Screening of databases, essentially based on X-ray determinations, often showed only very few structures with short H⋯F distances, for which reason the existence of such hydrogen bonds has been generally questioned. In contrast, crystallographic analyses have led to controversial conclusions. 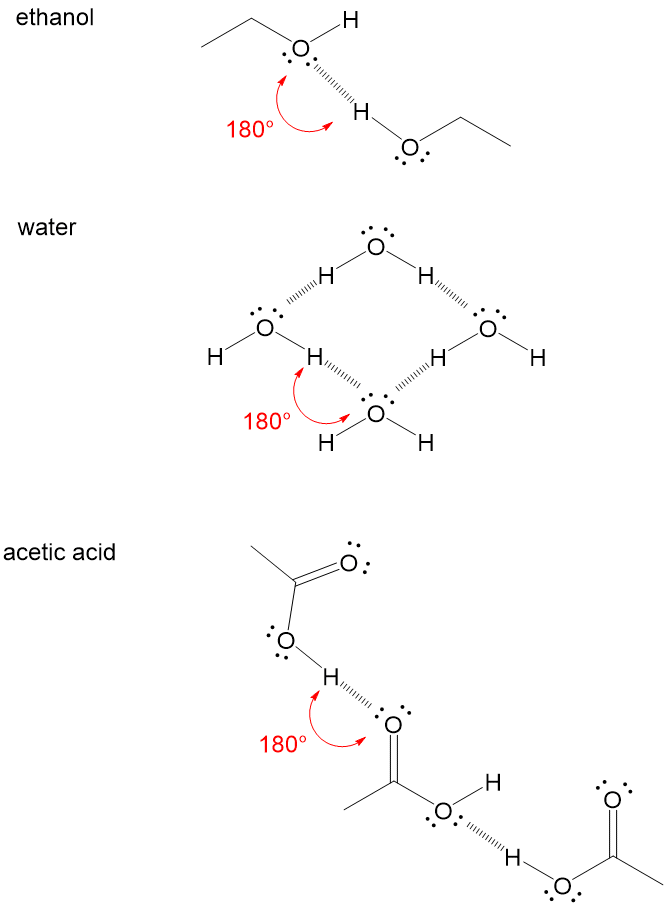
All computations point to dominating dispersive contributions, in particular for the very weak C–H⋯F interactions. 
Spectroscopic measurements unequivocally establish the presence of such hydrogen bonds, furnishing H⋯F distances as small as 2.02 Å, and X–H⋯F angles close to 180°, which generally agrees with most computational analyses. Simultaneous action of several X–H⋯F bridges can lead, even in polar media such as CDCl 3/CD 3CN, to binding constants of 70. 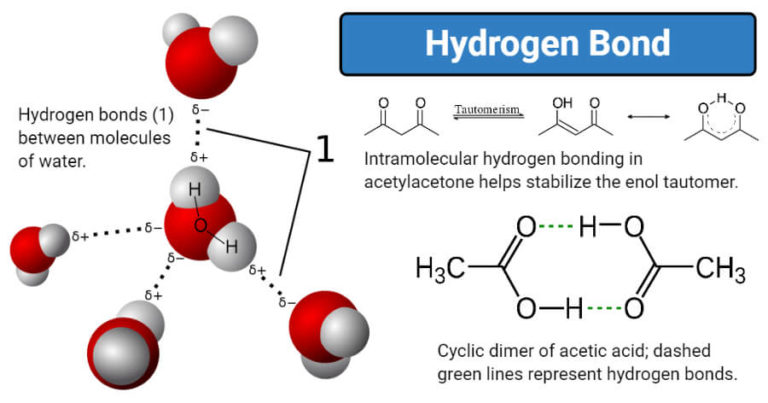
The measured Δ G values increase from primary to secondary to tertiary C–F moieties, ab initio calculations predict an increase from sp 3 to sp 2 to sp 1 carbon. Often overlooked experimental values for the free energy of hydrogen bond involving C–F as acceptor depend on the solvent in CCl 4 they amount with moderately acidic donors to Δ G = 6 kJ mol −1, much higher than with other halogens, placing fluorine as an acceptor between alkanes and arenes. Hydrogen bonds with organic fluorine are discussed on the background of an ongoing controversy, with an overview on the different methods of investigation, and with many examples, reaching from simple complexes to those involving nucleic acids. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
